1.
Canonical set of amino acids
1.1. What are amino acids
Living
beings are composed of various types of structures, including proteins. Amino
acids are part of the proteins in the form of separate units containing side chains.
Amino acids links in proteins can be allocated
in different ways. On
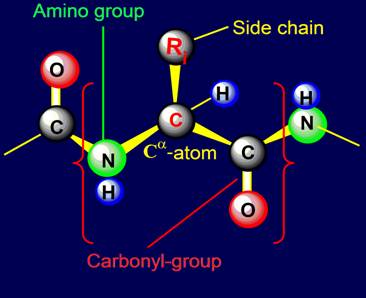
our website link is allocated as follows (Fig. 1):
|
Fig. 1. |
|
1. The link is in the brackets,
marked in with red color;
2. In the middle of the
link is Ca
- carbon atom, which bonds are in the shape of a tetrahedron;
3. On the left is an
amino-group of the link (green), on the
right - the carbonyl group (containing an oxygen atom in red);
4. Amino acids side chain
is attached to the top of Ca-carbon atom (indicated by the letters Ri The
letter ‘i’ can take different numerical
values. If the side chains are numbered from 1 to 20, the letter ‘i’
will be from 1 to 20.
5. The atom of hydrogen (blue
color) is also attached to the tetrahedral Ca-carbon atom.
1.1. How do the side chains of
amino acids look?
There are different classifications of
the side chains of amino acids. We will present a classification of the side
chains, which are often found in textbooks. This systematization divides the
side chains of amino acids into three groups:
1. Nonpolar
2. Weakly polar and polar
3. Cyclic
|
Nonpolar side chains |
|||||
|
Nonpolar side chains do not contain polar
groups and do not form hydrogen bonds |
|||||
|
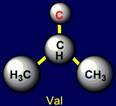
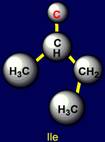
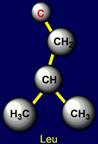
This group includes: Glycine (Gly), Alanine (Ala), Valine (Val), Isoleucine
(Ile), Leucine (Leu). Glycine has no side chain. Alanine has the shortest side chain,
leucine has the longest one. |
|
|
|
|
|
|
Glycine |
Alanine |
Valine |
Isoleucine |
Leucine |
|
|
Weakly polar and polar
side chains |
|||||
|
Typically, the side chains of this group form hydrogen bonds. These
bonds arise between two atoms through
atom of hydrogen, and are written as: XH ... Y. Hydrogen bonds are proton donor when a hydrogen atom attached to atom X and the proton acceptor, when an atom, which
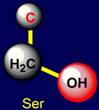
forms a hydrogen bond (Y), has no hydrogen atom. Proton donor hydrogen bonds form groups: C–OH of serine and threonine, O=C–OH - of aspartic acid and glutamic
acids, O=C–NH2- of asparagine
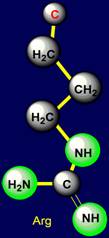
and glutamine, C–NH2 - lysine, HN= C–NH2 - of arginine, C–SH - of cysteine. Proton acceptor hydrogen bonds are formed with atoms O of the amino acids serine,
threonine, aspartic acid, glutamic acid, glutamine and aspragina, as well as the
S atom of methionine. |
|||||
|
Weakly
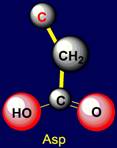
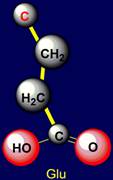
polar side chains concern: Polar are the following: Aspartic acid (Asp) and
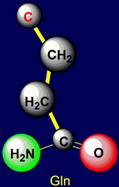
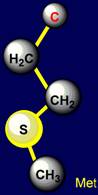
Glutamic acid (Glu), containing O=C–OH group, as well as Neutral, forming H-bonds are: Glutamine (Gln), with group O=C–NH2, and methionine (Met), containing a sulfur atom (group C–S) |
|
|
|
|
|
|
Serine |
Aspartic acid |
Glutamic acid |
Arginine |
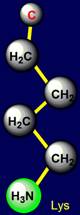
Lysine |
|
|
|
|
|
|
|
|
|
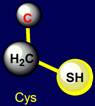
Сysteine |
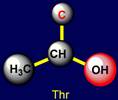
Threonine |
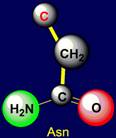
Asparagine |
Glutamine |
Methionine |
|
|
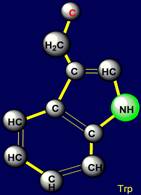
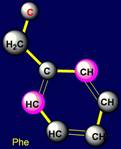
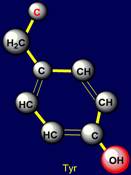
Cyclic side chains |
|||||
|
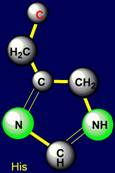
All cyclic amino acids, except proline, form hydrogen bonds.
Phenylalanine forms a weak hydrogen
bonds at the expense of CH groups, which are highlighted in pink in a
molecule. |
|||||
|
Cyclic side chains:
Phenylalanine (Phe), Tyrosine (Tyr), contain
6-membered ring |
|
|
|
|
|
|
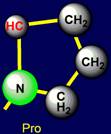
Proline |
Histidine |
Tryptophane |
Phenylalanine |
Tyrosine |
|
Knowing the structure of the side chains
of amino acids will help you in understanding the principles of constructing a
model of the spatial structure of the canonical set of amino acids. If you need
to remember one or another amino acid, one can look at this section.
The next stage of our work was to build
on the dodecahedron model of the structure of twenty formal elements. The model
described in Section 2.
Address
for connection: amino-acids-20@yandex.ru